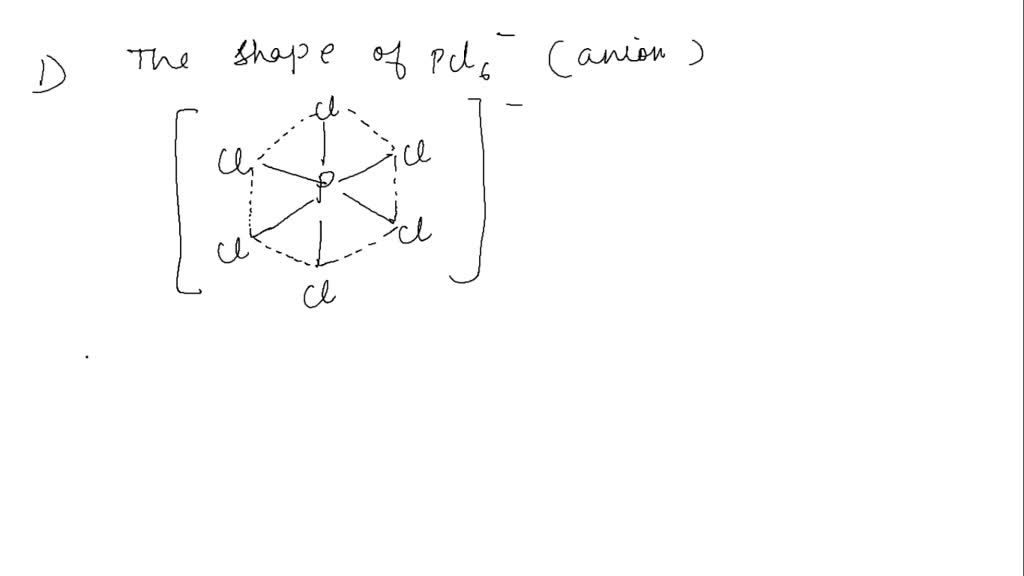
SOLVED: Answer the questions in the table below about the shape of the phosphorus hexachloride (PCl6-) anion. What word or two-word phrase best describes the shape of the PCl6- anion? What is
![In solid state PCl5 exist as an ionic solid [PCl4]+[PCl6]− hybridisation state of cation and anion respectively : In solid state PCl5 exist as an ionic solid [PCl4]+[PCl6]− hybridisation state of cation and anion respectively :](https://search-static.byjusweb.com/question-images/toppr_invalid/questions/124193_77117_ans.png)
In solid state PCl5 exist as an ionic solid [PCl4]+[PCl6]− hybridisation state of cation and anion respectively :

define hybridisation nd discuss d geometry of PCl6 nd SF6 moleculs on d basis of hybridisation - Chemistry - Chemical Bonding and Molecular Structure - 1536947 | Meritnation.com

![PCl5 is highly unstable and in solid state it exists as into [PCl4]+ and [ PCl6 PCl5 is highly unstable and in solid state it exists as into [PCl4]+ and [ PCl6](https://www.gkseries.com/blog/wp-content/uploads/2022/04/PCl5-is-highly-unstable-and-in-solid-state-it-exists-as-into-PCl4-and-PCl6-ions.-The-geometry-of-PCl6-is.png)


![PCl6]- - Phosphorus hexachloride PCl6]- - Phosphorus hexachloride](https://www.chemtube3d.com/images/gallery/inorganicsjpgs/PCl6_-.jpg)


![PDF] Regulation of Pcl6 and Pcl7 in a Glc7 pathway in Saccharomyces cerevisiae | Semantic Scholar PDF] Regulation of Pcl6 and Pcl7 in a Glc7 pathway in Saccharomyces cerevisiae | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/76c536bb9bbf4107997360edbceb5aab145e31a1/20-Figure1-1-1.png)


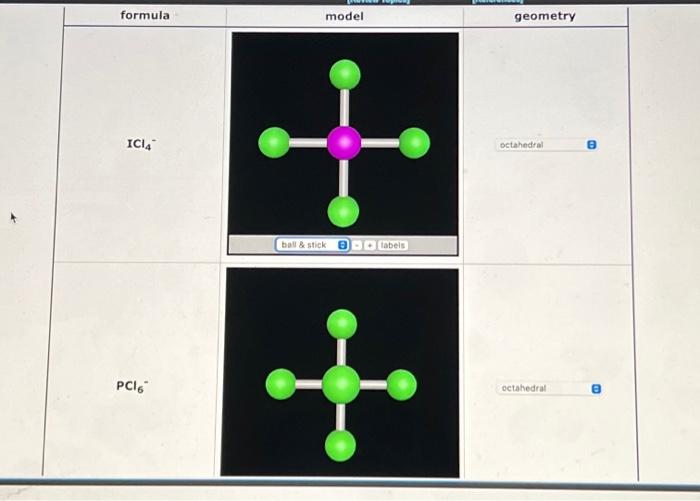
![What is the molecular structure of [PCl_6]^-, and what is the bond angle? | Homework.Study.com What is the molecular structure of [PCl_6]^-, and what is the bond angle? | Homework.Study.com](https://homework.study.com/cimages/multimages/16/pcl62958494712247179425.png)


![6. PCl6⊖ N=[N+]=[W] the actual shapes of the molecules. hole and not by .. 6. PCl6⊖ N=[N+]=[W] the actual shapes of the molecules. hole and not by ..](https://storage.googleapis.com/filo-question-diagrams/TgBirr_1697376970323638631_image_crop_58c9b052-d39d-4bfc-829e-716eaffe35ad7586088197733059965.jpg)



